Our most-documented batch. Janoshik ran a full panel from 21 to 24 April 2026: 99.741% HPLC, 20.92 mg retatrutide assay, heavy metals (As, Cd, Pb, Hg) not detected, TFA not detected, and bacterial endotoxin 1.449 EU/Vial via LAL. Endotoxin is the sterility benchmark pharma uses for release testing. Most research peptide sellers skip it. We publish it.
Batch Facts
- Product
- Retatrutide vial line (10mg and 40mg formats)
- Batch number
- RET-20-V-2604-001
- Molecule
- Retatrutide (LY-3437943), CAS 2381089-83-2
- Purity (HPLC)
- 99.741%
- Assay
- 20.92 mg retatrutide
- Heavy metals (As, Cd, Pb, Hg)
- Not detected
- Residual TFA
- Not detected
- Bacterial endotoxin (LAL)
- 1.449 EU/Vial
- Laboratory
- Janoshik Analytical
- Sample received
- 15 April 2026
- Analysis dates
- 21–24 April 2026
- Purity verify key
YCYA75M373NA- Heavy metals verify key
GHHWTCPUSEZ7- TFA verify key
NUA8B6XPAMWZ- Endotoxin verify key
E8ZFE17MI47D
Janoshik Reports

Purity + Assay
Common GLP-1 peptide blind test
99.741% HPLC · 20.92 mg assay
Verify key YCYA75M373NA

Heavy metals
As, Cd, Pb, Hg analysis
All four not detected
Verify key GHHWTCPUSEZ7
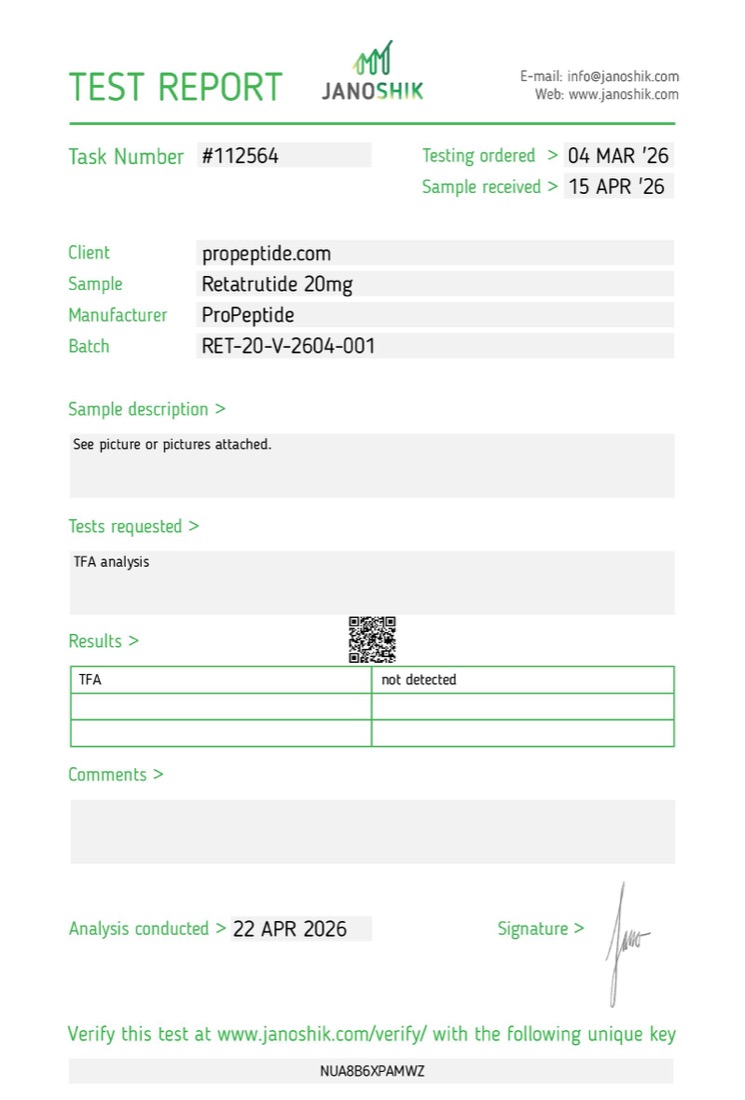
Residual solvent
TFA analysis
Not detected
Verify key NUA8B6XPAMWZ

Sterility benchmark
Bacterial endotoxin (LAL)
1.449 EU/Vial
Verify key E8ZFE17MI47D
What These Tests Prove
The vial-line batch ships with the most complete published panel: 99.741% HPLC purity, 20.92 mg assay, no heavy metals detected, no TFA detected, and 1.449 EU/Vial bacterial endotoxin via LAL. Each result has its own Janoshik verification key (Purity YCYA75M373NA, Heavy metals GHHWTCPUSEZ7, TFA NUA8B6XPAMWZ, Endotoxin E8ZFE17MI47D) so any single number can be audited against the original report.
What these tests do not prove: LAL detects bacterial endotoxin contamination but is not a sterility certification. The reports cover the tested lot at the test date and do not warrant shelf life beyond the date stamp or correct handling after dispatch.
Common Questions
What is batch RET-20-V-2604-001?
Retatrutide vial line, covering both 10mg and 40mg formats. Janoshik ran a full panel 21–24 April 2026: 99.741% HPLC, 20.92 mg retatrutide assay, heavy metals (As, Cd, Pb, Hg) not detected, TFA not detected, bacterial endotoxin 1.449 EU/Vial.
What does 1.449 EU/Vial mean?
EU is endotoxin units, measured via the LAL (Limulus Amebocyte Lysate) assay. 1.449 EU/Vial is the bacterial endotoxin load found in the vial. Endotoxin is the sterility benchmark pharma uses for release testing. Most research peptide sellers skip it; this batch publishes the number.
Why test for TFA?
TFA (trifluoroacetic acid) is a common residual solvent from peptide synthesis. HPLC purity does not see it. Testing separately confirms it is not present. For this batch, TFA was not detected.
How do I verify each Janoshik report?
Go to
janoshik.com and paste the relevant verification key. Purity:
YCYA75M373NA. Heavy metals:
GHHWTCPUSEZ7. TFA:
NUA8B6XPAMWZ. Endotoxin:
E8ZFE17MI47D. Each returns the original report off Janoshik's server.
What product ships with batch RET-20-V-2604-001?
Where to go next




